1. Anti-fatigue agent
2. Anti-tumor and cancer preventative (multiple means of cancer prevention are cited)
3. Anti-aging elixir
4. Anti-oxidant agent
5. Hypotensive (lowers blood pressure)
6. Hypoglycemic (lowers bood sugar levels having an anti diabetic effect)
7. Immunostimulant (multiple catagories cited)
8. Antilipidemic – lowers blood fat levels
9. Anti-obesity – promotes weight loss
10. Antherosclerotic – prevents hardening of arteries
11. Cardioprotective
12. Antiviral
13. Antibiotic – modulates and prevents bacterial infections)
14. Anti-depressent
15. Anxyolytic – anti-anxiety effect
16. Anti-Alzheimerian – helps prevent dementia
17. Anti-arthritic
18. Antiperidontic – prevents gum disease
19. Antistenoporosis – helps prevent loss of bone mass
20. Antiallergenic – helps prevent allergic reactions
21. Antiseborric – prevents dandruff
22. Anticalculictic – helps prevent kidney stones
23. Antipyrectic – lowers fever
24. Anti-Parkinsons
25. Antidiahrreal
26. Antineuralgic – reduces nerve pain
27. Antivertigo – reduces dizziness
28. Antiglaucomic
29. Anticataract
Gamma tocotrienol
Gamma tocotrienol (123) seems to be a telomerase activator according to Terraternal, so that Annatto, a plant with a pod that opens to reveal a kiss heavy with fruit rich in gamma tocotrienol, is an interesting new telomerase activator.
Mucuna puriens
Mucuna Pruriens HGH Secreatagogue
Mucuna Pruriens, commonly known as velvet bean, contains a significant
amount of naturally occurring L-dopa, an amino acid that converts into
dopamine. Mucuna Pruriens has long been used in traditional Ayurvedic
Indian medicine for Parkinson Disease.
In a randomized, controlled,
double blind clinical trials, researchers has found that the natural
source of L-dopa from Mucuna Pruriens possess advantages over
conventional L-dopa preparations in the long term management of
Parkinson Disease. Dopamine is an essential component of our body and
it's required for proper functioning of the brain.
Research has discovered that the body converts the amino acid tyrosine
into L-dopa; L-dopa is then converted into dopamine.
Without the
neurotransmitter dopamine to serve a damping effect on neural
transmissions, muscles become tense and tremble.
L-dopa may be useful for improving attention deficit hyperactivity
disorder (ADHD) by improving sleep disorder. ADHD is a chronic,
neurologically based syndrome characterized by restlessness,
distractibility and impulsivity. Many patients with ADHD experience
sleep disorder that may cause them to be inattentive and hyperactive due
to sleep deprivation.
According to a study released during the American
Academy of Neurology, patients taking Levadopa showed marked
improvement in their sleep disorder, behavior, and mental acuity.
Researchers argue that there may be a common link -- a dopaminergic
deficiency in the brain that causes both the sleep disorders and the
ADHD.
Early pharmacological studies, published in 1927 and 1930
respectively, proved (in the rabbit) that L-dopa exerted significant
effects on glucose metabolism (causing marked hyperglycemia) and on
arterial blood pressure. Powdered Mucuna Pruriens seeds (contains 3-4% L
Dopa) were given to normal and diabetic rabbits. In normal group 0.5, 1
and 2 g/kg of M. Pruriens significantly decreased the blood glucose
levels while in diabetic rabbits only 1 and 2 g/kg body weight caused a
significant fall. High levels of trace elements like manganese, zinc,
and others were found in these seeds. Therefore, it is conceivable that
M. Pruriens seeds contain hypoglycemic principles, may be both organic
and mineral, which seem to act indirectly by stimulating the release of
insulin and/or by a direct insulin-like action.
In 1977 issue of SCIENCE, researchers reported that L-dopa increased
mean lifespans by up to 50% when fed to experimental rats at a dose of
40 to 80mg per gram of food.
In a 1979 research, L-dopa almost totally
reversed the slowness of motion and lack of coordination shown by aged
rats (24 to 27 months). Aged rats could not swim well for more than a
few minutes and their swimming lacked coordination compared to control
young rats, and after several minutes they would repeatedly sink. All of
these problems disappeared after treatment with L-Dopa.
These research
present strong evidence that regular treatment with L-Dopa could have
anti-aging effects and therefore lead to greater vigor in the old.
A group of Urologist from Southern France used apomorphine (Dopamine
activator) for the treatment of erectile dysfunction. When animals have
probes implanted that let them self-stimulate the pleasure pathway
[dorsal raphe - ventral tegmental area – nucleus accumbens
(NA)/prefrontal cortex (PFC)], they will often stimulate dopamine
release into the NA/PFC, rather than eat or sleep, until they starve to
death. This provides a strong evidence that participation of
dopaminergic system in the control of sexual function.
Dopamine is a
precursor to adrenaline. Dopaminergic networks are associated with
"pleasure and movement centers" and are thought to control mood,
motivation and reward. Many rewards and pleasures can ‘trip the dopamine
pleasure switch’, including sex, food, hugging, kissing, etc.
Experimental data suggest that the release of dopamine are present at
all stages of sexual behavior in rodents.
It is likely that dopamine can
trigger penile erection by acting on oxytocinergic neurons located in
the paraventricular nucleus of the hypothalamus, and perhaps on the
pro-erectile sacral parasympathetic nucleus within the spinal cord.
Mucuna Pruriens has also been popular among athletes as L- Dopa contains
natural secretagogues which may support the body's ability to stimulate
the natural release ootropin, is a
protein made up of 191 amino acids which is secreted by the pituitary
gland and coordinates normal growth and development. Receptors which
respond to hGH exist on cells and tissues throughout the body. The most
obvious effect of HGH is on linear skeletal development. But the
metabolic effects of hGH on muscle, the liver, and fat cells are
critical to its function. The increased dopamine levels also optimize
the production of other hormones, including testosterone, leading to
increased sex drive and improved sexual performance for both men and
women, beneficial in stimulating muscle growth, as well as burning fat
from fat cells
L-Dopa provides a large number of health benefits:
HELPS REGENERATION OF ORGANS (HEART, KIDNEY, LIVER, LUNGS)
HELPS DRAMATICALLY STRENGTHENED IMMUNE SYSTEM
HELPS IMPROVED SLEEP (PROMOTES DEEP SLEEP)
HELPS REDUCE BODY FAT & DECREASED CELLULITE
DECREASES WRINKLES
IMPROVES SKIN TEXTURE AND APPEARANCE
PROMOTES INCREASE BONE DENSITY & REVERSAL OF OSTEOPOROSIS
HELPS INCREASE LEAN MUSCLE MASS
HELPS IMPROVE MOOD AND SENSE OF WELL BEING
HELPS ENHANCE LIBIDO & SEXUAL PERFORMANCE
HELPS INCREASE ENERGY LEVELS
HELPS IMPROVE CHOLESTEROL PROFILE
Source - http://www.calcompnutrition.com/
Tankyrase
(35) Tankyrase (Tankyrase 1,
telomeric PARP)
[GeneCards/Tankyrase,
Links,
Images,
Papers,
Patents,
Books,
WikiGenes/Tankyrase,
JL Mergny, JF Rois, et al., 2002;
Links/supplements promoting Tankyrase;
Images/Tankyrase molecule;
Links/Tankyrase 1,
Images,
Papers,
Patents,
Books;
Links/the Tankyrase 1 promoter,
Images,
Papers,
Patents,
Books;
Links/supplements promoting Tankyrase 1;
sources;
toxicity;
tankyrase 1
plasmid expression vectors,
Tankyrase 1 cDNA].
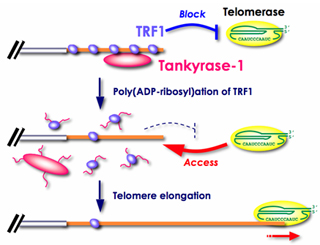
Tankyrase, a telomeric poly(ADP-ribose) polymerase (PARP), "acts as a positive regulator of telomere enlongation in vivo, apparently by inhibiting TRF1 [Index]. In general, PARP catalyzes the formation of poly(ADP-ribose) onto a protein acceptor using NAD+ as a substrate in response to DNA damage.
Long-term overexpression of tankyrase 1 in telomerase-positive human cells results in a gradual and progressive enlongation of telomeres. See "Overexpression of tankyrase 1 in telomerase-positive cells releases TRF1 from telomeres, resulting in telomere elongation." [2004], and H. Seimiya (2006),
The telomeric PARP, tankyrases, as targets for cancer therapy, British Journal of Cancer, 2006, 94, 341-345 [Figure 2].
Tankyrase 1 uses NAD as a stubstrate in poly(ADP-ribosylation) of TRF1, so NAD supplements [Images] may be useful when applying Tankyrase 1 to encourage hTERT transcription to activate telomerase.
Furthermore, nicotinamide
(niacinamide),
a form of Vitamin B3,
has been shown to boost
poly(ADP-ribose) polymerase
levels (Tammy M. Jackson, et. al., 1995).
Nicotinamide was used
to elevate PARP levels in experimental animals at 1 gram/kg dosage,
and was useful for upregulating NAD+ in the heart
at 500 mg/kg. "Phosphorylation
of tankyrase 1
results in upregulation of its intrinsic PARP activity."
(H. Seimiya,
2006). Phosphorylation is accomplished by insulin stimulation.
Tankyrase 1
is serine-phosphorylated by MAP kinases,
enhancing its telomeric PARP activity.
(Sbodio,
Lodish, and Chi, 2002).
"...Tankyrase is quantitatively phosphorylated on
certain serine residues
by MAP kinase upon stimulation with
insulin,
PDGF, and EGF."
(Chi
and Lodish, 2000).
Tankyrase 1 has more than 1 function in the cell. "Tankyrase 1 is a novel signaling target of mitogen-activated protein kinase (MAPK); it is stoichiometrically phosphorylated upon insulin stimulation. Phosphorylation enhances the poly(ADP-ribose) polymerase activity of tankyrase 1..." (Nai-Wen Chi and Harvey F. Lodish, 2000).
Phosphorylation allows tankyrase 1 to transduce MAPK signaling into poly(ADP-ribosyl)ation of effector proteins such as TRF1.
See upregulating tankyrase 1 mRNA transcription [Papers]. "TRF1 is a specific negative regulator of telomere length and its poly(ADP-ribosylation) by tankyrase 1 leads to the loss of telomere association and subsequent degradation by the ubiquitin/proteasome pathway (Chang et al. 2003, Smith et al. 1998)." (W.N.Keith and A.E.Bilsland, Targeting Telomerase: Therapeutic Options for Cancer Treatment, in K. Lenhard Rudolph, 2008, p.269.)
Perhaps adding an extra gene for tankyrase 1 with Zinc Finger Nuclease technology would be life-extending, if it did not much encourage carcinogenesis. Alternatively, tankyrase 1 plasmid expression vectors may be useful. See promoting tankyrase transcription. Evidently, going for a strong insulin spike after a bodybuilding workout (by using a specially formulated post-workout shake) can phosphorylate tankyrase, improving its performance as a telomerase activator. Furthermore, insulin itself may have this effect.
Gymnema Sylvestre (400-500 mg with a postworkout shake < 30 min after exercise) stimulates insulin secretion. Dextrose may be taken at 25-50 grams to spike insulin, along with whey protein or whey hydrolysates after a bodybuilding workout. Insulin secretion may also be improved by Fenugreek seeds, Fenugreek Extract [Images], or 4-hydroxyisoleucine [Images]. Tankyrase action on TRF1 can probably be promoted using Gymnema Sylvestre [Images] to produce insulin, phosphorylating Tankyrase 1 to increase its telomerase activation activity.
I note that a "tanky" race of men keeps its insulin high.
See TwinLabs
Creatine Nitrate3 Fuel.
See also Antisense
inhibition of Bcr-Abl/c-Abl synthesis promotes telomerase activity
and upregulates tankyrase in human leukemia cells,
FEBS letters,
2004, vol. 564, no1-2, pp. 73-84 [12 page(s) (article)].
Tankyrase 1 has a short reported half-life of just 2 hours.
Tankyrase 1 is overexpressed in human breast cancer,
so that novel
tankyrase inhibitors
(MST-312,
MST-295, and
MST-199) have been developed to function as
telomerase inhibitors for
application to anticancer therapy.
Furthermore, overexpression of
tankyrase 2, a related protein, results
in cell death (references from
H.
Seimiya, 2006). I add that there are also tankyrase-related proteins incorporating certain domains
of tankyrase 1 that perform like tankyrase 1 for purposes of opening telomere loops. There are also
nucleic acids (recombinant DNA molecules) that can bind to the acidic domain of a
TRF1. See De Lange, et al.,
TRF1 Binding Protein, Methods of Use Thereof,
US Patent
No. 6,277,613 B1, Aug. 21, 2001.
It seems clear that extra tankyrase 1 might be expressed by
supplying a DNA plasmid for transfection into the cell that is designed to encode tankyrase 1. Plasmids
with inducible promoters may be used, or perhaps
Plasmids of suitable half-life may be designed.
See Anderson, et al.,
US Patent 5,399,345.
Other compounds besides niacinamide might be used to improve NAD substrate levels for tankyrase 1 reactions with TRF1, and targeted phosphorylation of tankyrase 1 using substances besides insulin may be envisioned, perhaps vanadyl sulphate.
Also note that the effect of tankyrase 1 is different
on long telomeres (> 20 kbp) than on short telomeres,
where it acts to remove TRF1 for telomerase access (De Lange, et al, 2001). This because long telomeres
seem to support a coiled higher-order structure in the presence of sufficient TRF1. See
Smith, S., I. Giriat, A. Schmitt, and T. de Lange. (1998),
Tankyrase,
a poly(ADP-ribose) polymerase at human telomeres,
Science 282:1484-1487.
See also Cook, B. D., J. N. Dynek, W. Chang, G. Shostak, and S. Smith (2002), Role for the related poly(ADP-Ribose) polymerases tankyrase 1 and 2 at human telomeres, Mol. Cell. Biol, 22:332-342. Also of revelance is Ye J.Z., and de Lange, T. (2004), TIN2 is a tankyrase 1 PARP modulator in the TRF1 telomere length control complex, Nature Genetics, 36, 618-623. See also Nai-Wen Chi and Harvey F. Lodish (2000), Tankyrase Is a Golgi-associated Mitogen-activated Protein Kinase Substrate That Interacts with IRAP in GLUT4 Vesicles, Journal of Biological Chemistry, December 8, 2000, 275, 38437-38444. This is a reference for the phosphorylation of tankyrase 1 resulting in the upregulation of its activity. "In insulin-stimulated adipocytes, tankyrase 1 is phosphorylated at serine residues by the mitogen-activated protein kinase pathway." - After H. Seimiya, op.cit., referencing (Chi and Lodish, 2000), op.cit. Note that "...Tankyrase is quantitatively phosphorylated on certain serine residues by MAPK upon stimulation with insulin, PDGF, and EGF." (Chi and Lodish, 2000).
The phosphorylation of tankyrase 1 enhances its poly(ADP-ribo)sylation activity on TRF1 telomere loop closure protein, allowing t-loops to open for access by the telomerase holoenzyme. For application of tankyrase wisdom to cancer, see Hiroyuki Seimiya, Yukiko Muramatsu, Tomokazu Ohishi, and Takashi Tsuruo (2005), Tankyrase 1 as a target for telomere-directed molecular cancer therapeutics, Cancer Cell, Vol.7, issue 1, January 2005, pp.25-37. Also see Susan Smith and Titia de Lange (2000), Tankyrase promotes telomere enlongation in human cells, Current Biology, vol 10, no. 20, 1299-1302 with Susan Smith and Titia de Lange, TRF1 Binding Protein, Methods of Use Thereof, US Patent 6,277,613 B1. "Tankyrase does not contain a nuclear localization signal (NLS) and transfected tankyrase is excluded from the nucleus unless it is co-transfected with TRF1.... (An NLS can be added to its amino terminus to produce nuclear-localized tankyrase (FN-tankyrase))... Tankyrase releases TRF1 from telomeres in a reaction that depends on its PARP domain".
Doxycyclin
Presence or absence of telomere growth can depend on the presence of doxycyclin
[Links/doxycyclin,
Images,
Papers,
Patents,
Books;
Links/doxycyclin and telomere growth,
Images,
Papers,
Patents,
Books].
Tankyrase, a telomeric poly(ADP-ribose) polymerase (PARP), "acts as a positive regulator of telomere enlongation in vivo, apparently by inhibiting TRF1 [Index]. In general, PARP catalyzes the formation of poly(ADP-ribose) onto a protein acceptor using NAD+ as a substrate in response to DNA damage.
Long-term overexpression of tankyrase 1 in telomerase-positive human cells results in a gradual and progressive enlongation of telomeres. See "Overexpression of tankyrase 1 in telomerase-positive cells releases TRF1 from telomeres, resulting in telomere elongation." [2004], and H. Seimiya (2006),
The telomeric PARP, tankyrases, as targets for cancer therapy, British Journal of Cancer, 2006, 94, 341-345 [Figure 2].
Tankyrase 1 uses NAD as a stubstrate in poly(ADP-ribosylation) of TRF1, so NAD supplements [Images] may be useful when applying Tankyrase 1 to encourage hTERT transcription to activate telomerase.
Tankyrase 1 has more than 1 function in the cell. "Tankyrase 1 is a novel signaling target of mitogen-activated protein kinase (MAPK); it is stoichiometrically phosphorylated upon insulin stimulation. Phosphorylation enhances the poly(ADP-ribose) polymerase activity of tankyrase 1..." (Nai-Wen Chi and Harvey F. Lodish, 2000).
Phosphorylation allows tankyrase 1 to transduce MAPK signaling into poly(ADP-ribosyl)ation of effector proteins such as TRF1.
See upregulating tankyrase 1 mRNA transcription [Papers]. "TRF1 is a specific negative regulator of telomere length and its poly(ADP-ribosylation) by tankyrase 1 leads to the loss of telomere association and subsequent degradation by the ubiquitin/proteasome pathway (Chang et al. 2003, Smith et al. 1998)." (W.N.Keith and A.E.Bilsland, Targeting Telomerase: Therapeutic Options for Cancer Treatment, in K. Lenhard Rudolph, 2008, p.269.)
Perhaps adding an extra gene for tankyrase 1 with Zinc Finger Nuclease technology would be life-extending, if it did not much encourage carcinogenesis. Alternatively, tankyrase 1 plasmid expression vectors may be useful. See promoting tankyrase transcription. Evidently, going for a strong insulin spike after a bodybuilding workout (by using a specially formulated post-workout shake) can phosphorylate tankyrase, improving its performance as a telomerase activator. Furthermore, insulin itself may have this effect.
Gymnema Sylvestre (400-500 mg with a postworkout shake < 30 min after exercise) stimulates insulin secretion. Dextrose may be taken at 25-50 grams to spike insulin, along with whey protein or whey hydrolysates after a bodybuilding workout. Insulin secretion may also be improved by Fenugreek seeds, Fenugreek Extract [Images], or 4-hydroxyisoleucine [Images]. Tankyrase action on TRF1 can probably be promoted using Gymnema Sylvestre [Images] to produce insulin, phosphorylating Tankyrase 1 to increase its telomerase activation activity.
Other compounds besides niacinamide might be used to improve NAD substrate levels for tankyrase 1 reactions with TRF1, and targeted phosphorylation of tankyrase 1 using substances besides insulin may be envisioned, perhaps vanadyl sulphate.
See also Cook, B. D., J. N. Dynek, W. Chang, G. Shostak, and S. Smith (2002), Role for the related poly(ADP-Ribose) polymerases tankyrase 1 and 2 at human telomeres, Mol. Cell. Biol, 22:332-342. Also of revelance is Ye J.Z., and de Lange, T. (2004), TIN2 is a tankyrase 1 PARP modulator in the TRF1 telomere length control complex, Nature Genetics, 36, 618-623. See also Nai-Wen Chi and Harvey F. Lodish (2000), Tankyrase Is a Golgi-associated Mitogen-activated Protein Kinase Substrate That Interacts with IRAP in GLUT4 Vesicles, Journal of Biological Chemistry, December 8, 2000, 275, 38437-38444. This is a reference for the phosphorylation of tankyrase 1 resulting in the upregulation of its activity. "In insulin-stimulated adipocytes, tankyrase 1 is phosphorylated at serine residues by the mitogen-activated protein kinase pathway." - After H. Seimiya, op.cit., referencing (Chi and Lodish, 2000), op.cit. Note that "...Tankyrase is quantitatively phosphorylated on certain serine residues by MAPK upon stimulation with insulin, PDGF, and EGF." (Chi and Lodish, 2000).
The phosphorylation of tankyrase 1 enhances its poly(ADP-ribo)sylation activity on TRF1 telomere loop closure protein, allowing t-loops to open for access by the telomerase holoenzyme. For application of tankyrase wisdom to cancer, see Hiroyuki Seimiya, Yukiko Muramatsu, Tomokazu Ohishi, and Takashi Tsuruo (2005), Tankyrase 1 as a target for telomere-directed molecular cancer therapeutics, Cancer Cell, Vol.7, issue 1, January 2005, pp.25-37. Also see Susan Smith and Titia de Lange (2000), Tankyrase promotes telomere enlongation in human cells, Current Biology, vol 10, no. 20, 1299-1302 with Susan Smith and Titia de Lange, TRF1 Binding Protein, Methods of Use Thereof, US Patent 6,277,613 B1. "Tankyrase does not contain a nuclear localization signal (NLS) and transfected tankyrase is excluded from the nucleus unless it is co-transfected with TRF1.... (An NLS can be added to its amino terminus to produce nuclear-localized tankyrase (FN-tankyrase))... Tankyrase releases TRF1 from telomeres in a reaction that depends on its PARP domain".
Doxycyclin
Subscribe to:
Comments (Atom)